Trans-Epithelial Electrical Resistance (TEER)
Epithelial and endothelial cells form barriers in the body. The strength and integrity of these barriers can be assessed via measurements of the electrical resistance across the cell layer in vitro, called TEER.
TEER is a well-established method of evaluating and monitoring epithelial tissue in a non-destructive assay. In particular, the confluence of the monolayer is quickly determined. The confluence can be tracked and monitored in real-time as the TEER measurement will rise as the gaps in the monolayer close.
TEER is often used with epithelial and endothelial cells in a monolayer as a strong indicator of cell barrier integrity and permeability. These indicators are often used in vitro with evaluating transport of drug chemicals or drug screening assays.
How does TEER work:
Impedance technology is used to measure TEER. A small AC current is passed from one electrode to another. TEER measures how much of this electrical signal is blocked by the cellular layer, thereby quantifying barrier integrity. Because impedance is non-invasive and label-free, the barrier can be continuously monitored for minutes, hours, or even days without disturbing the cellular biology.
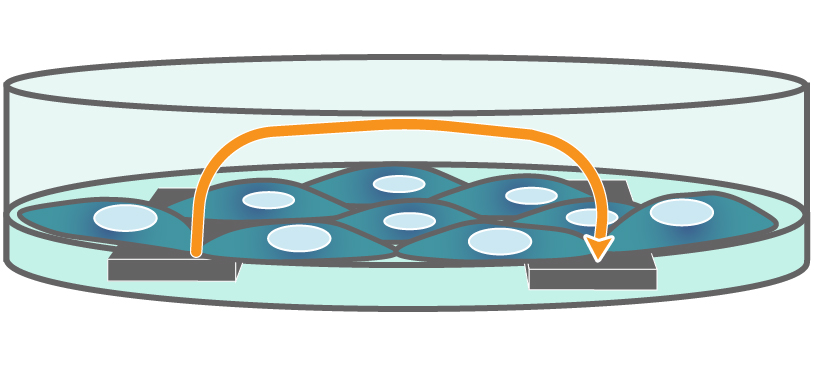
Electrodes embedded in the cell culture substrate at the bottom of each well detect small changes in the impedance of current flow. Barriers, such as tight junctions between cells, resist current flow, leading to an increase in TEER measurements of barrier function.
Traditionally, TEER is measured by placing two “chopstick” style electrodes on either side of a transwell insert with a confluent cell layer. Manual methods, where the electrodes are placed well-by-well, are highly labor intensive. Even automated TEER methods are typically constrained to lower throughputs, like 24 well plates but now systems allow measurements of up to 96 wells simultaneously.
Common biological models evaluated with TEER:
>> Blood-brain barrier
>> Gastrointestinal tract
>> Pulmonary models
>> Organ-on-a-chip
The biological models are often assessed with TEER in order to evaluate drug or chemical transport or diffusion.
Measuring frequencies of electrical resistance:
Traditional TEER measurements require a fully confluent cell layer to accurately measure barrier function. In contrast, newer systems track coverage and TEER simultaneously and continuously by measuring impedance at multiple frequencies.
Measuring impedance at low frequency is highly sensitive to the intercellular barrier formed by tight junctions and the paracellular barrier formed by cell membranes. On the other hand, measurements at higher frequency can be used to quantify coverage of the well bottom. In other words, low frequencies are sensitive to “what” cells are there, whereas high frequencies are sensitive to “how many” cells are there.
“The Teer assay is a game-changer in cell barrier research.”
The Teer assay is a vital tool for unraveling the mysteries of cellular barriers and advancing our understanding of health and disease in the easiest way possible. We utilize the Maestro Edge system with the Impedance Module to measure TEER in colonic epithelial cells cultured with various bacteria in vitro. Recently, we demonstrated that B. adolescentis bacteria impair epithelial barrier integrity in patients with irritable bowel syndrome (IBS).
- Hadar Bootz-Maoz. Bar Ilan University, Ramat Gan, Israel

Transcript of the What is TEER video
Epithelial tissue is a major building block of many organs, like the skin, lungs, liver, kidney, and digestive tract. Specialized epithelial cells called endothelium comprise the inner lining of blood vessels.
While they may take a variety of forms, epithelial tissues serve an important biological function forming protective barriers throughout the body and regulating what gets in and out. Tightly packed cells attach together with junction proteins that form selectively permeable barriers. When these barriers are disrupted chemically, through injury, or disease, this regulation breaks down leading to a host of adverse effects. Measuring in vitro barrier integrity with TEER is a widely accepted method to quantify and model barrier function.
TEER stands for trans-epithelial electrical resistance. Traditionally TEER is measured by manually placing two chopstick-style electrodes on each side of a confluent cell layer. A low frequency current is applied and the resistance of the barrier is measured. The easier the current flows between the cells the lower the TEER value. This method is typically low throughput and variability can arise from electrode placement, changing environmental factors, and more. A fully confluent layer is required to accurately measure barrier function. By integrating electrodes into the well consistent placement is ensured with the Maestro Z reducing variability. Up to 384 wells are measured simultaneously, hands-free. A fully controlled environment ensures barrier function can be stably monitored for minutes, hours, or days without disturbing the cells automatically.
By measuring at multiple frequencies the Maestro Z can simultaneously track TEER and confluence. Low frequency measurements are sensitive to the barrier properties, while high frequency measurements detect cell confluence over the well bottom. By measuring both, differences in confluence are controlled and accounted for in your analysis. In this example, both Calu-3 and A549 cell lines are of epithelial origin and both cell lines reach full coverage within 24 hours. However, only the Calu-3 line expresses tight junction proteins so it produces a much stronger barrier which stabilizes only after the cells reach confluence. Without measuring confluence it is difficult to know when TEER will be stable. TEER may be used for disease modeling and drug development.
The cystic fibrosis transmembrane conductance regulator or, CFTR protein, is an anion channel that regulates secretion and absorption in multiple tissues. Discovered for its role in cystic fibrosis where mucous secretion may be disrupted, the channel is stimulated by cyclic AMP production. Upon stimulation, CFTR opens to allow ion transport across the cell membrane resulting in a rapid and substantial drop in TEER. A selective CFTR inhibitor blocks this decrease in a dose-dependent manner confirming CFTR as the underlying mechanism of the response. How could TEER help your research? Learn more about TEER and the Maestro Z at axionbio.com/TEER





