TEER
TEER
What is TEER-Based Barrier Analysis?
Trans-epithelial/endothelial electrical resistance (TEER) is a functional measure of barrier integrity across epithelial or endothelial cell layers. TEER assays quantify how effectively a cell monolayer restricts ion flow, providing a direct readout of tight junction formation and permeability. Maestro Z enables continuous TEER monitoring in multiwell plates, allowing researchers to track barrier development and disruption in real time.
Why Does Real-Time Barrier Monitoring Matter?
Barrier tissues play essential roles throughout the body, including in the lung, gut, vasculature, and blood–brain barrier. Disruption of barrier integrity is a hallmark of inflammation, infection, drug-induced toxicity, and disease. Traditional TEER methods are often low-throughput and manual, limiting kinetic insight. Maestro Z overcomes these limitations by delivering automated, hands-free TEER measurements across 96 wells simultaneously.
Key Advantages of Maestro Z TEER Technology
Label-free, noninvasive impedance recording for long-term barrier monitoring
Easy, hands-free workflow from minutes to days
96-well CytoView-Z plates enable high-throughput assays and microscopy compatibility
SIntegrated environmental controls for acute and chronic studies
Reliable, simultaneous well-to-well measurements collected each minute
Automated experiment management via barcode tracking
The TEER assay is a game-changer in cell barrier research. The TEER assay is a vital tool for unraveling the mysteries of cellular barriers and advancing our understanding of health and disease in the easiest way possible.
How TEER Works on Maestro Z
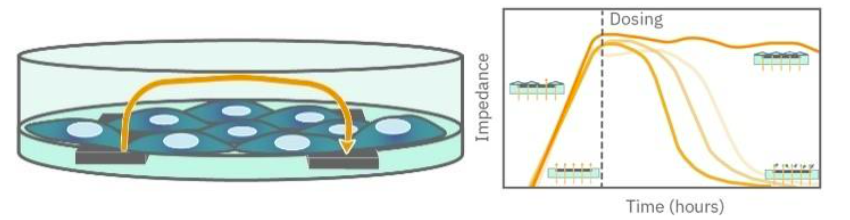
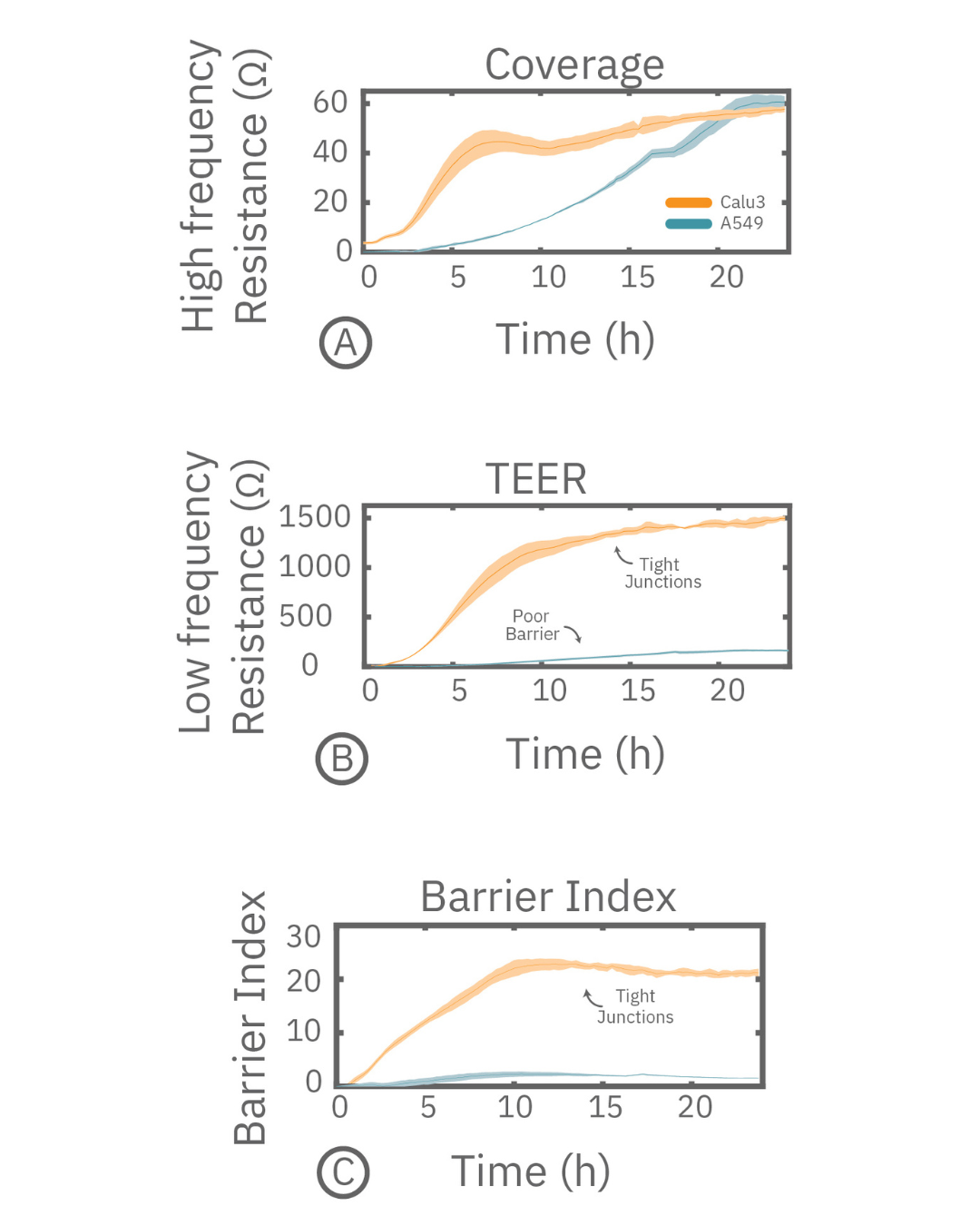
TEER assays measure the electrical resistance across a cell layer. As epithelial or endothelial cells form tight junctions, they restrict ion movement, increasing resistance and indicating stronger barrier integrity. Maestro Z simultaneously measures cell coverage and barrier resistance using multi-frequency impedance, enabling accurate normalization of TEER signals.
How Maestro Z TEER Assays Work: Step-by-Step Workflow
Maestro Z TEER assays integrate seamlessly into standard barrier culture workflows, enabling continuous, high-throughput monitoring with minimal hands-on steps.

Barrier cell seeding and monolayer formation
Epithelial or endothelial cells are cultured and seeded into CytoView-Z electrode plates using standard protocols. No dyes or invasive reagents are required.

System loading and environmental stabilization
Plates are placed into Maestro Z, which maintains controlled temperature and environmental conditions for acute or long-term TEER studies.

Continuous real-time TEER and coverage monitoring
Impedance is measured continuously to track both monolayer coverage and barrier resistance as tight junctions develop or are disrupted.

Quantitative data analysis and barrier integrity readouts
Signals are converted into TEER metrics that reflect permeability, junction strength, and dynamic responses to drugs or biological stimuli.
How to Interpret TEER Data
Maestro Z TEER assays provide kinetic insight into barrier biology by capturing both gradual barrier maturation and rapid permeability changes. By measuring TEER continuously, researchers can distinguish true barrier formation from simple cell coverage and detect transient disruptions, recovery dynamics, and mechanism-specific responses that are missed by endpoint measurements.
Technology Differentiators FAQ
Manual TEER methods require low-throughput, electrode-by-electrode measurements. Maestro Z automates TEER acquisition across 96 wells simultaneously.
No. TEER measurements are label-free and noninvasive, preserving barrier biology for downstream analysis.
Continuous measurement enables detection of transient disruptions and recovery dynamics that endpoint methods miss.
Yes. CytoView-Z plates and automated acquisition support high-throughput permeability and safety screening workflows.
Multi-frequency impedance enables normalization of TEER to cell coverage, improving consistency across wells and experiments.