Image
Featured Application
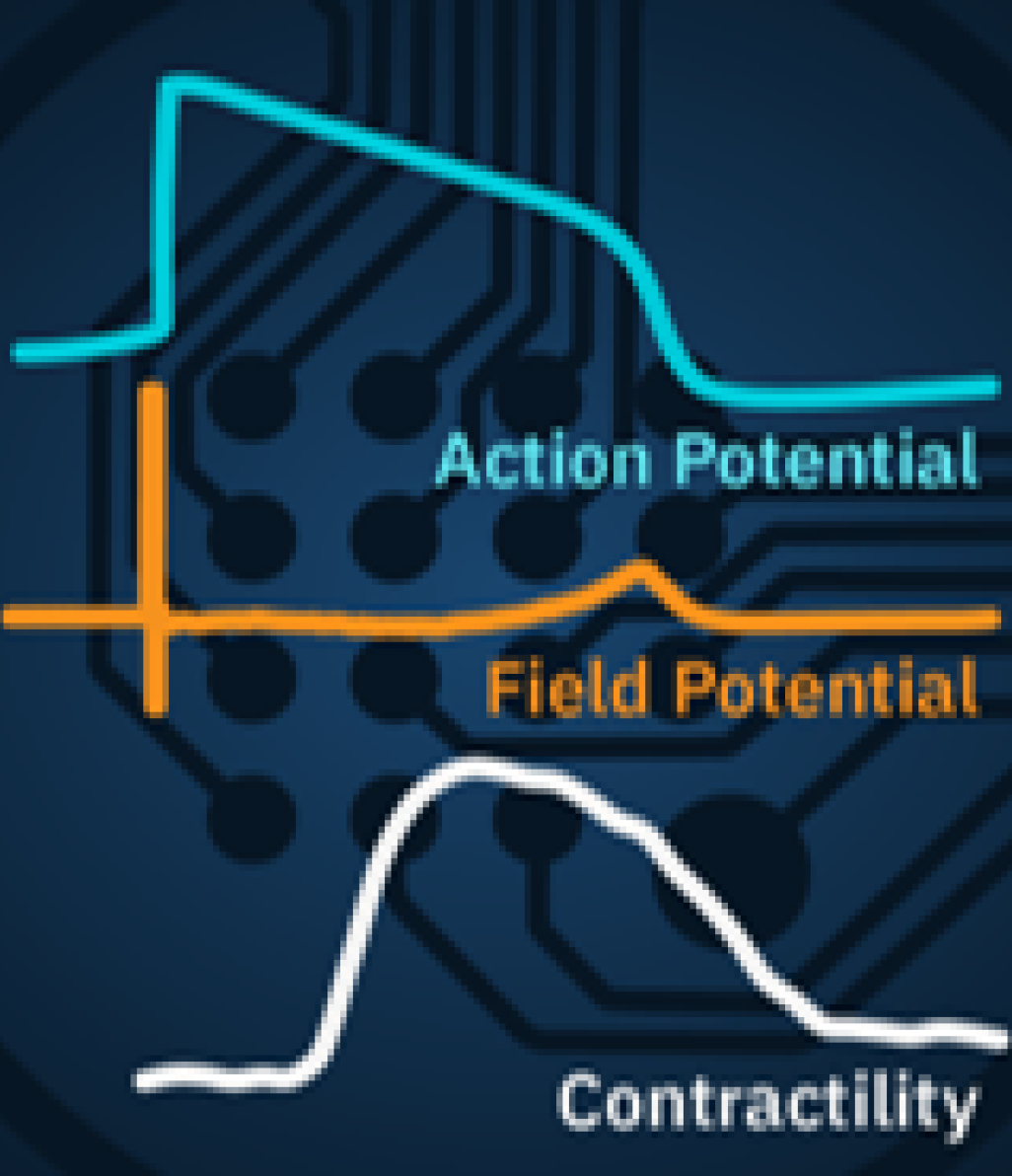
Cardiac Characterization
Characterize cardiomyocyte function in vitro to accelerate cardiovascular disease research, drug development, and safety testing.
Learn More
→