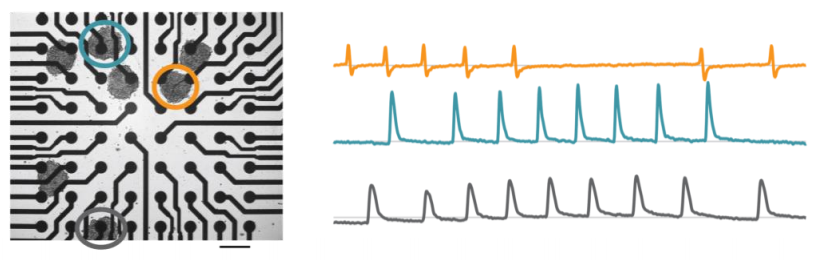
See more with array-based contractility
What is High-Res contractility?
The Maestro Pro and Edge offer high resolution contractility by sampling from many locations across the whole culture. When cardiomyocytes are plated over a microelectrode array, they form a spontaneously beating syncytium. Every beat is characterized by a propagating electrical impulse, measured as the field potential, that triggers a mechanical contraction. As the cells contract and relax, cells change shape and alter the coverage over the electrodes. This change can be measured by each electrode in the array as a change in impedance, or contractility.
Detect changes in cardiomyocyte contractility
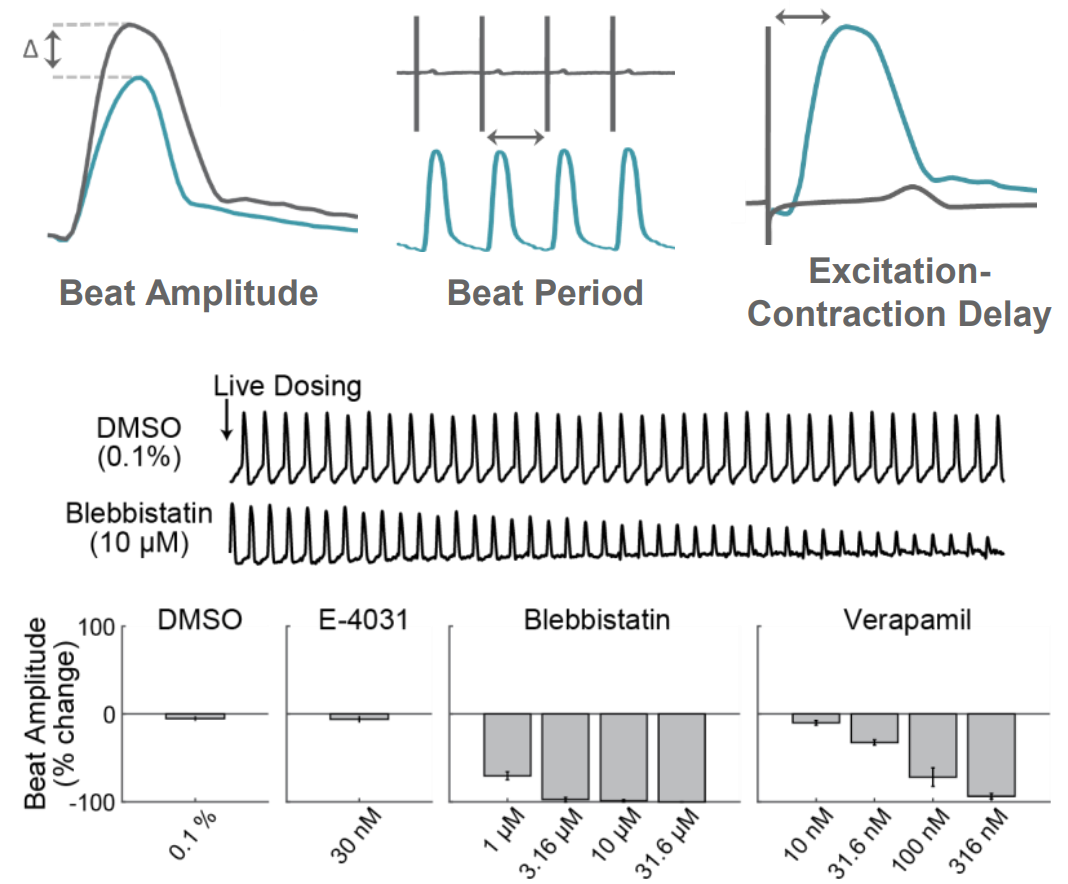
AxIS Navigator provides measures of both electrode and well-wide contractility, including Beat Amplitude, Beat Period, and Excitation-Contraction Delay. In the example below, cardiomyocytes were live dosed with Blebbistatin, a myosin inhibitor, and Verapamil, a calcium channel inhibitor. For both compounds, increasing concentrations resulted in a reduction in contractility beat amplitude, in contrast to no change with DMSO or E-4031.
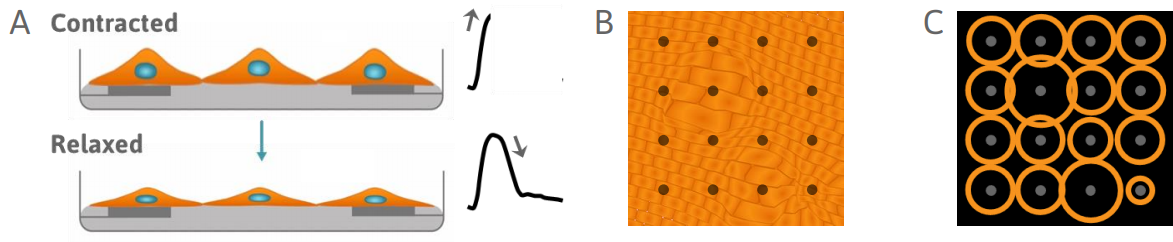
The Maestro Advantage
>> Track local changes in contractility
>> Enable contractility with 3D constructs, such as spheroids and organoids
>> Elicit mature phenotypes with only 48 hours of chronic pacing
>> Measure cultures with wide variations in cell coverage and attachment
>> Mature iPSC-CMs and measure contractility, field potentials, propagation, and action potentials in a single system
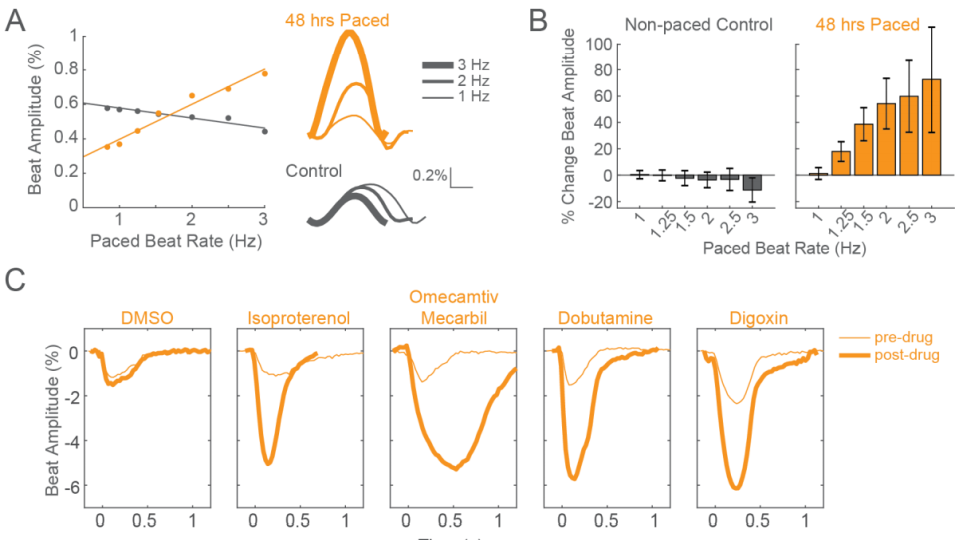
Faster Functional Maturity
Elicit mature contractility phenotypes after only 48 hours of chronic pacing
Human induced pluripotent stem cell-derived cardiomyocytes (iPSC-CMs) are limited by functional immaturity, including immature calcium handling and contractility function. Physical conditioning of iPSC-CMs via electrical or mechanical stimulation facilitates maturation as measured by a positive force-frequency relationship and detection of positive inotropes. Current in vitro protocols require 2-4 weeks of chronic pacing. Using high-res contractility and local electrical stimulation, the Maestro elicits functionally mature phenotypes after only 48 hours.
Contractility from 3D constructs
High throughput 3D culture electrophysiology
Three-dimensional in vitro cell models, such as spheroids and organoids, more accurately recapitulate the multicellular organization and structure of in vivo tissues, making them powerful for disease modeling, developmental biology, and drug safety. Recording from an array of microelectrodes enables contractility measures from one or more 3D cardiac spheroids in a well. A large electrode would smear these signals or not detect them at all. Thus, array-based contractility not only enhances traditional applications, but uniquely enables advanced contractility applications.