Cardiac MEA
Cardiac MEA
What is Cardiac MEA
Cardiac MEA (microelectrode array) technology is a label-free method for recording electrical activity in heart cells. Tiny electrodes embedded in the culture surface detect electrical signals from cardiomyocytes, allowing continuous, noninvasive monitoring of cardiac function in real time. This technology provides insights into cardiomyocyte function, rhythm, and drug response in 2D and 3D cell models
Why Cardiac MEA matters
Traditional cardiac assays rely on indirect, low resolution, or endpoint measurements that fail to capture electrical function, rhythm changes, and time-dependent drug effects. Cardiac MEA technology addresses this gap by providing direct, physiologically relevant measurements of cardiac electrophysiology, supporting safer drug development, improved cardiotoxicity screening, and more predictive disease models.
Unique Capabilities of Cardiac MEA Technology
Noninvasive, real-time recording of cardiac electrical activity without dyes or labels
High temporal and spatial resolution to capture beat rate, rhythm, and conduction patterns
Long-term monitoring of cardiomyocyte function, including maturation and drug response
Multiwell, scalable formats suitable for comparative and screening studies
Quantitative, reproducible electrophysiology data aligned with translational cardiac metrics
An easy-to-use system that offers a complete solution without the need for additional software or electrophysiological or bioinformatic skills. When you identify a proper way to plate your cells of interest in the MEA multi-well plates, measurements and data analysis is straightforward and the results are reproducible and of high-quality.
Scientific Principles Underlying Cardiac MEA Technology
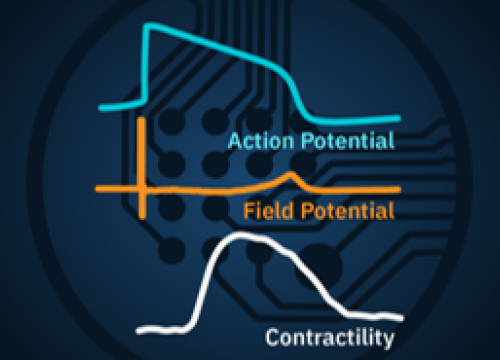
When cardiomyocytes are cultured on top of an MEA, they attach and connect to form a spontaneously beating sheet of cells, called a syncytium. When one cardiomyocyte fires an action potential, the electrical activity propagates across the syncytium causing each cell to fire and then contract. The electrodes detect each individual action potential and contraction, as well as the propagation of this activity across the array.
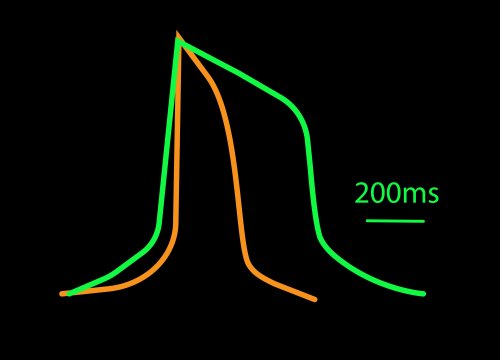
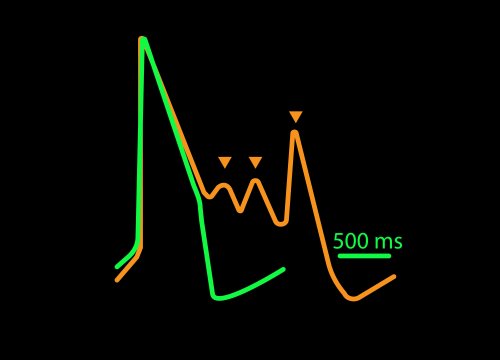
The propagating electrical signal is detected by the electrodes as an extracellular field potential. The field potential derives from the underlying cardiac action potential, but more closely resembles a clinical electrocardiogram (ECG) signal. The initial depolarization phase is seen as a sharp spike, similar to the QRS complex, and the slow repolarization is seen as a small slow spike, like a Twave. The time from the depolarization to repolarization is termed the field potential duration (FPD) and is a key metric in predictive cardiotoxicity screening assays.
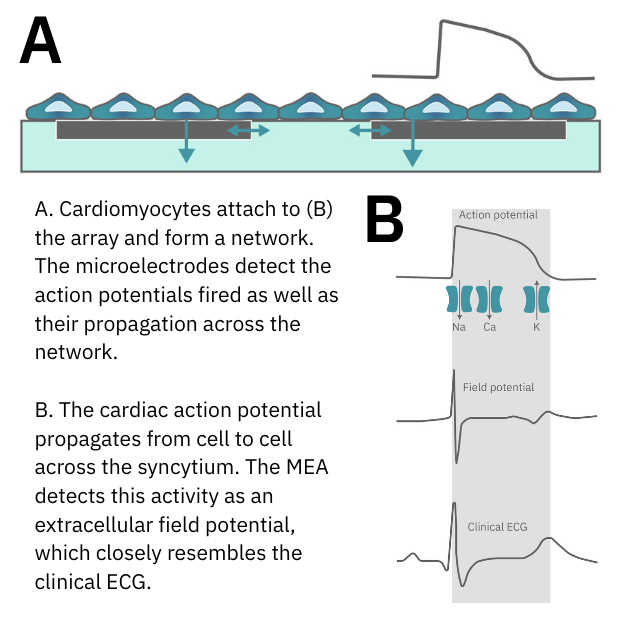
Key Components of MEA Technology
While most systems record the cardiac field potential, the Maestro Pro and Edge MEA systems can measure local extracellular action potentials (LEAP). LEAP enhances the coupling between microelectrodes and cardiomyocytes, allowing for detailed metrics such as rise time, action potential duration (APD), and automated early afterdepolarization (EAD) detection.
 Measure what matters
Measure what matters
Indirect measures are often used to infer cardiac activity. However, calcium imaging fails to capture subtle changes in Na+ channel functionality, and protein marker expression levels often do not correlate well with cell models.
 Capture the complete timeline
Capture the complete timeline
With real-time data recording, the Maestro Pro detects key parameters of cardiac functionality, and cell growth, death, and attachment.
 Record with Maestro
Record with Maestro
Record the four key measures of functional cardiac performance, label-free and in real-time in every well of the multi-well plate: [1] Action Potential; [2] Field Potential; [3] Propagation; and [4] Contractility.
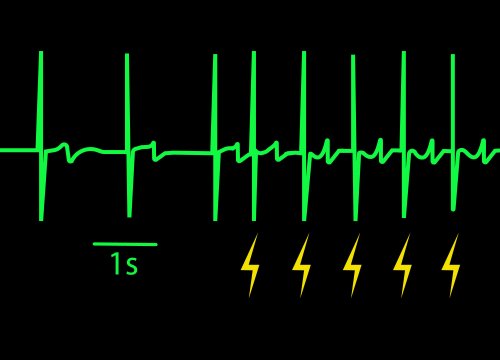
 LEAP (Local Extracellular Action Potential) Induction
LEAP (Local Extracellular Action Potential) Induction
Optional feature to enhance signal quality by promoting coupling between electrodes and cardiomyocytes.
How to perform a Cardiac MEA assay

Cardiomyocytes are cultured and seeded into MEA plates. Cells attach and form a functional beating network.
The MEA plate is placed into the Maestro and is ready for automated recording.
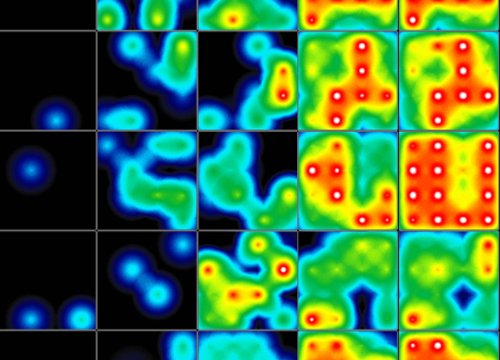
As cardiomyocytes beat, the Maestro continuously records signals from each electrode, capturing beat timing, rhythm, and how electrical activity propagates across the cell network in real time.

Recorded signals are processed into waveforms and quantitative metrics.
Interpreting Cardiac MEA data
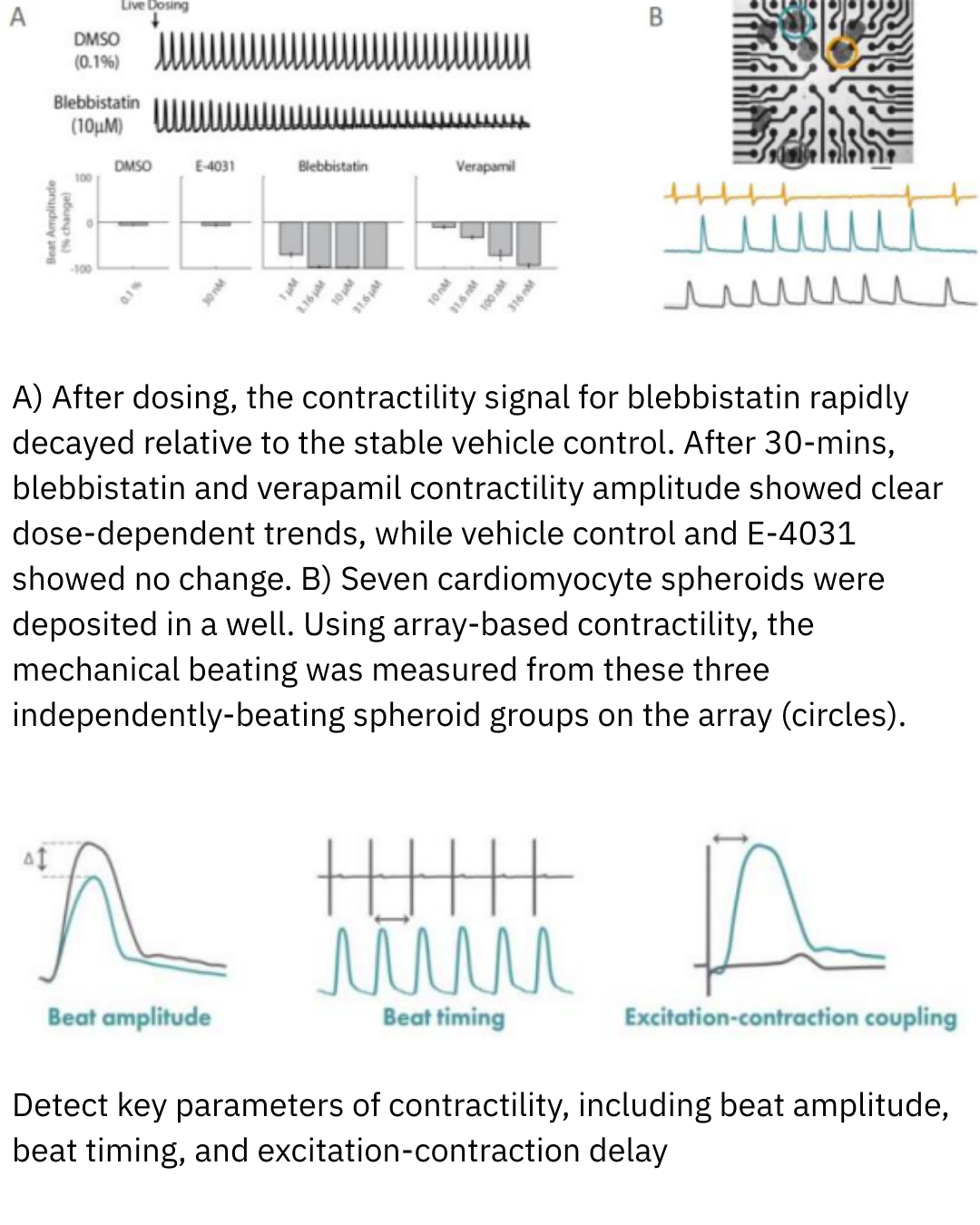
Cardiac MEA on the Maestro MEA system generates data by recording the extracellular electrical signals produced as cardiomyocytes fire and propagate action potentials across the cell network. Each electrode detects local electrical activity, and these signals are combined to create waveforms and spatial maps of cardiac function. The resulting data quantify key features, including beat rate, rhythm regularity, conduction velocity, and repolarization timing.
These measurements provide a direct, functional view of cardiac electrophysiology, revealing how heart cells behave individually and as a synchronized tissue. Broadly, Cardiac MEA data indicate cardiac health, functional maturity, and response to external stimuli, enabling researchers to detect arrhythmias, assess drug-induced risk, and compare physiological differences across conditions with high sensitivity and translational relevance.
How Cardiac MEA compares to other methods
| What are the competing technologies? | Maestro Cardiac MEA | Optical/Imaging-Based Assays | Patch Clamp |
| How do they compare in sensitivity? | Maestro Detects subtle changes in beat rate, rhythm, and conduction across networks | Opical is Limited by dye intensity or signal-to-noise ratio | Patch clamp Measures single-cell ionic currents with high precision |
| How do they compare in reproducibility? | Automated, standardized recordings across wells | Variable; sensitive to staining, imaging conditions | Operator-dependent; high skill required for consistency |
| How do they compare in throughput? | Multiwell formats enable scalable experiments | Moderate; imaging throughput limited by setup | Low; manual or semiautomated |
| How do they integrate/multiplex with other assays? | Compatible with multimodal assays, software analysis, and downstream workflows | Standalone or requires custom integration | Standalone; data integration can be complex |