Cardiac LEAP
Cardiac LEAP
What is LEAP?
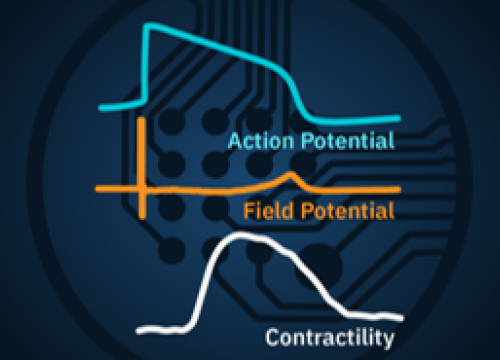
LEAP technology enables noninvasive, label-free monitoring of the cardiac action potential in a high-throughput, real-time format. LEAP can be used for quantification of action potential morphology, repolarization irregularities such early after depolarizations (EADs), and arrhythmic risk factors such as triangulation.
Why LEAP matters?
The LEAP signal accurately reflects the shape and duration of the underlying action potential. The large signal allows for automated detection and classification of arrhythmic events, such as notched EADs, rolling EADs, or ectopic beats. LEAP also provides metrics not available from the field potential, such as rise time and triangulation.
Unique Capabilities of LEAP Technology
Automated APD and EAD detection for high-throughput drug screening
Predicting arrhythmic risk for cardiac safety and cardiotoxicity testing
Characterization of action potential morphology in hiPSC-cardiomyocytes
Studying the effects of genetic manipulation in cardiac electrophysiology
Comparing cardiac biology in healthy and diseased states
The LEAP assay addresses an important gap in the field, namely providing a non-invasive solution in recording high quality action potentials from cardiac cells using a high throughput format. The LEAP assay may be a game changer.
Scientific Principles Underlying LEAP Technology
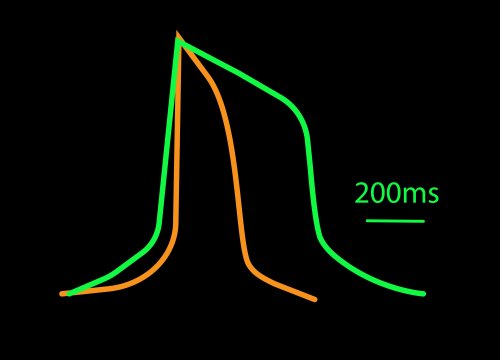
LEAP stands for local extracellular action potential. The theory behind LEAP is similar to that of patch clamp, where the recorded signal amplitude is proportional to the sealing resistance between the electrode and the cell.
In contrast to a field potential signal, LEAP induction increases the coupling between the cells and electrode, enabling the measurement of a much larger action potential signal. The increased cell-electrode coupling is stable for 10-20 minutes or longer, allowing extracellular monitoring of the cardiac action potential without disrupting the underlying biology with dyes or invasive electrodes.
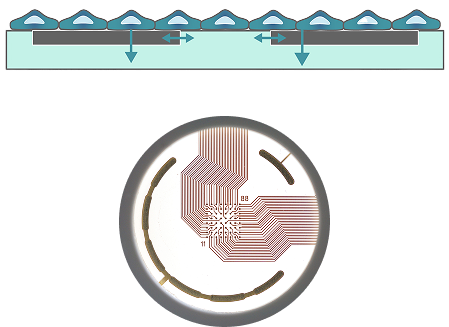
How to perform a Cardiac LEAP assay

Cardiomyocyte Seeding and Plate Preparation
Coat MEA plate with fibronectin, following by plating of cardiomyocytes.
MEA System Loading
Dock the plate in your Maestro Pro.
MEA Recording Initialization
Create your plate map, add any required compounds and start LEAP induction.

Advanced Cardiac Data Analysis
Review LEAP endpoints in the Axion’s analysis software.
Interpreting LEAP data
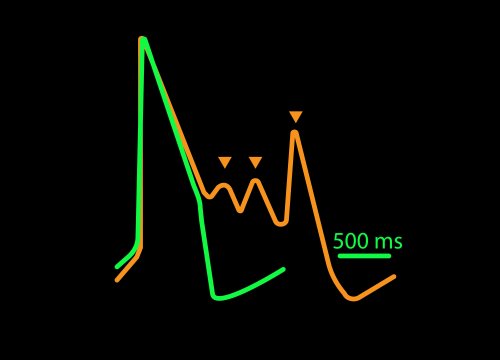
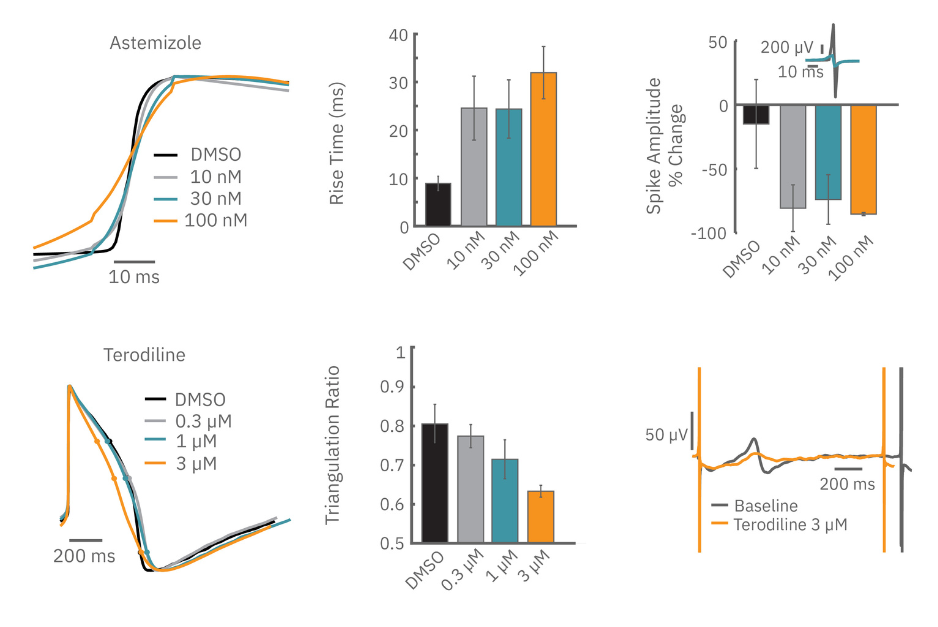
Sodium channel blockers cause a detectable change in spike amplitude. At higher doses though, the spike amplitude can become too small to detect. However, rise time prolongation in the LEAP signal can still be easily measured even at high doses. Similar effects can be seen at high doses of hERG blockers when hERG block begins to impact the resting membrane potential.
Similarly, compounds, such as terodiline, that induce triangulation can flatten the field potential repolarization feature, referred to as the “T wave”. The resulting broad, small amplitude T wave is difficult to detect and quantify. In contrast, triangulation is readily detectable and quantifiable in the LEAP signal.
How LEAP compares to other methods
| What are competing cardiac technologies? | Maestro Cardiac MEA | Patch-clamp | Voltage Dyes |
| How do they compare in sensitivity? | Medium-High, captures AP morphology, but measures only extracellular membrane voltage | High, direct intracellular membrane voltage measurement | High, but signals are changes in relative fluorescence, not absolute voltages) |
| How do they compare in reproducibility? | Good, label-free and noninvasive | Variable, dependent on operator skill | Moderate, dependent on dye loading uniformity |
| How do they compare in throughput? | High, can measure up to 96 wells | Very Low, performed on once cell at a time | Medium, limited by microscope capabilities |
| How do they integrate/multiplex with other assays? | High, can measure LEAP, FP, conduction velocity, and contractility | Low, targeted primary at electrophysiology | Medium, can be combined with Ca2+ imaging |