Optogenetics
Optogenetics
What is Optogenetics?
Optogenetics is a technique that involves the use of light to control cell function. Cells are first genetically modified to express light-sensitive ion channels, called opsins. Then, light can be used to activate the opsin. The most well-known opsins are light-gated ion channels that can control the excitability of the cell membrane.
Why Optogenetics Matters
By delivering precise, time-locked stimulation, optogenetics enables researchers to interrogate circuit behavior, model disease-related dysfunction, and evaluate therapeutic interventions with high temporal and spatial resolution.
Unique Capabilities of Optogenetic Technology
Enables precise control of genetically defined cell populations using light
Provides millisecond-scale temporal precision to stimulation and inhibition
Supports high-throughput, parallel assays in multiwell formats
Allows reversible and programmable modulation for repeatable experiments
Lumos is a snap to start working with right off the bat. The software makes it really easy to start controlling different lights with different colors and pulse patterns within a couple minutes. By hooking up a light meter under the plate to an oscilloscope, I could see that the light is controlled very precisely (microsecond precision) with really high max intensity reaching the bottom of the well. Perhaps the best thing for me was that the plate/lid design prevents light targeted to one well from bleeding over or reaching adjacent wells, which is pretty important for experiments.
Scientific Principles Underlying Optogenetics Technology
Since the first microbial opsin was introduced into mammalian neurons in 2005, many different opsins have been used to control the excitability of electroactive cells such as neurons and cardiomyocytes. Each opsin is sensitive to a specific wavelength range, or color of light, and induces a precise biological event.
Channelrhodopsin (ChR2), for example, is activated by blue light. When ChR2 opens, positive cations (like sodium and calcium), flow into the cell, depolarizing or exciting the cell. In contrast, halorhodopsin and archaerhodopsin both inhibit cell excitability by hyperpolarizing the cell in response to yellow or green light, respectively. With optogenetics, you can turn on and off cells like a light switch.
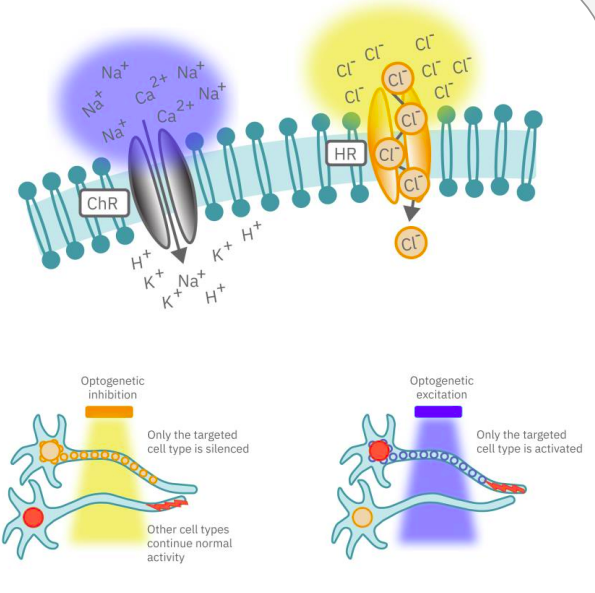
Key Components of Optogenetics Technology
The timing of these light-activated events is fast, facilitating highly precise control. First generation opsins, such as channelrhodopsin, open and close in milliseconds, ideal for kicking off an action potential. Second generation opsins have fine-tuned kinetics for even faster, more precise control or slower, longer-lasting inhibition. Unlike electrical stimulation, which excites all nearby cells, optical stimulation can be finely targeted to the cells expressing the opsins responsive to a narrow band of light wavelengths.
 Precise control of targeted cells
Precise control of targeted cells
Optogenetics enables selective activation/inhibition of genetically defined cell populations using light, allowing researchers to turn specific cells “on” or “off.”
 High temporal resolution
High temporal resolution
Light activated opsins respond on the scale of milliseconds, giving fast, time-locked control of cellular activity.
 Spatial specificity
Spatial specificity
Unlike electrical stimulation that affects all nearby cells, optogenetics targets only those expressing light-sensitive proteins, enabling fine spatial discrimination.
 Reversible, non-invasive modulation
Reversible, non-invasive modulation
The effects of light stimulation via the Lumos are reversible and do not require permanent physical interventions, simplifying dynamic studies of cell function.

How to perform an Optogenetic assay

Add virus to cells.

Coat MEA plate with fibronectin, following by plating of cells.

Apply optical stimuli to cells and perform experiment.
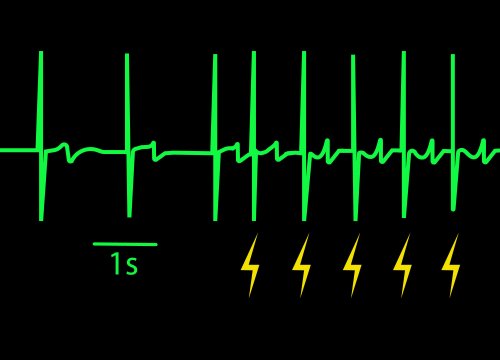
Interpreting Optogenetic data
ChR2 and Chronos neurons were dosed with either a proconvulsant (picrotoxin,10µM), antiepileptic (carbamazepine, 30µM). Neural networks dosed with picrotoxin showed a sustained response to high frequency blue light stimulation (2ms pulses at 40Hz for 1s) in which firing did not return to zero between each pulse, resulting in a higher cumulative spike count across the 40 pulses. In contrast, carbamazepine reduced the response, resulting in a lower cumulative spike count.
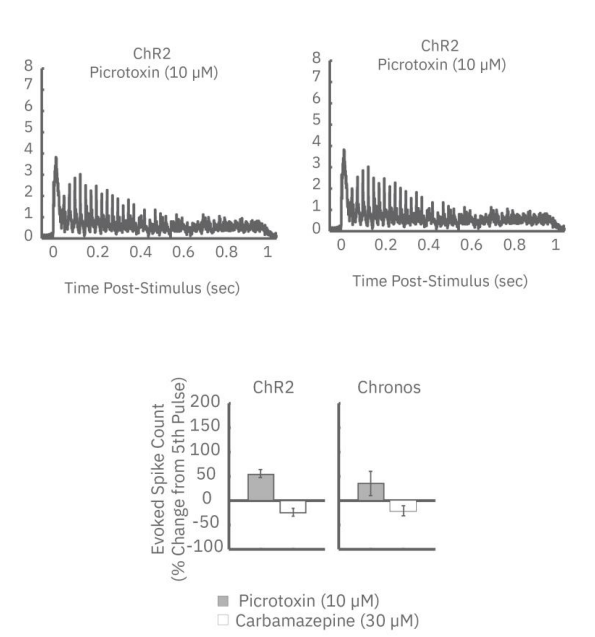
How Optogenetics compares to other methods
| What are competing stimulation technologies? | Optogenetics | Electrical Stimulation | Chemogenetics | Magnetic Stimulation |
| How do they compare in sensitivity? | High, can stimulate only genetically targeted cells expressing light-sensitive proteins | Low, affects all excitable cells near the electrode | Moderate, targets cells expressing designer receptors, but lacks temporal control | Low – influences larger regions |
| How do they compare in temporal precision? | High, millisecond-scale control with rapid light pulses | High, electoral pulses can be precise, but can also trigger unintended activity | Low, pharmacological activation has slower onset/offset | Moderate, temporal resolution is constrained by slower physiological responses |
| How do they compare in reproducibility? | High, repeated light stimuli produce consistent responses | Variable, reproducibility can be affected by activation of variable cell populations | Moderate, reproducible at the population level but subject to variability in drug dosing | Moderate, reproducible, but biological responses can vary |
| How do they compare in throughput? | High, Lumos delivers independent light to multi-well plate simultaneously | High, Maestro MEA can record from up to 96 wells simultaneously | Moderate, throughput depends on how many samples can be processed in parallel | Low, targets one region at a time and does not scale easily to mult-well |